UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
Pursuant to Rule 13a-16 or 15d-16
of the Securities Exchange Act of 1934
February 9, 2021
Commission File Number: 001-39522
COMPASS PATHWAYS PLC
(Exact name of registrant as specified in its charter)
| England and Wales | 2834 | Not applicable | ||||||||||||||||||||||||||||||||||||||||||
(State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification Number) | ||||||||||||||||||||||||||||||||||||||||||
COMPASS Pathways plc
3rd Floor
1 Ashley Road
Altrincham
Cheshire WA14 2DT
United Kingdom
Tel: +1 (646) 905-3974
(Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F. Form 20-F ☒ Form 40-F ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ☐

COMPASS Pathways Plc
COMPASS Pathways Plc (the “Company”) will be conducting meetings with investors beginning on February 9, 2021. As part of these meetings, the Company will deliver the slide presentation furnished to this report as Exhibit 99.1 and which is incorporated herein by reference.
The information in this report included as Exhibit 99.1 and incorporated herein by reference shall not be deemed “filed” for the purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or
otherwise subject to the liabilities of that section. It may only be incorporated by reference in another filing under the Exchange Act or the Securities Act of 1933, as amended, if such subsequent filing specifically references the information furnished pursuant to this report.

EXHIBITS
| Exhibit | Description | |||||||
| 99.1 | ||||||||

SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| COMPASS PATHWAYS PLC | |||||||||||||||||
| Date: February 9, 2021 | By: | /s/ George Goldsmith | |||||||||||||||
Name: Title: | George Goldsmith Chief Executive Officer | ||||||||||||||||

© COMPASS Pathways 2021 Transforming mental health care COMPASS Pathways plc February 2021

© COMPASS Pathways 2021 Disclaimer This presentation has been prepared by COMPASS Pathways plc (“we”, “us”, “our”, “COMPASS”, “COMPASS Pathways”, or the “Company”) and contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Except for statements of historical fact, information contained herein constitutes forward-looking statements and includes, but is not limited to, expectations regarding our success in pre- clinical studies or clinical trials, the success of COMP360 in treating treatment-resistant depression, planned expansion into additional indications, the benefits of psilocybin therapy, the success of our future patent strategy, our ability to reach a large number of patients and differentiate our commercial offering and the success of our strategic partnerships. Words such as, but not limited to, “look forward to”, “believe”, “expect”, “anticipate”, “estimate”, “intend”, “plan”, “would”, “should” and “could”, and similar expressions or words, identify forward- looking statements. Such forward-looking statements necessarily involve known and unknown risks and uncertainties, which include, without limitation, the severity of the impact of the COVID-19 pandemic on the Company’s business, including on pre-clinical and clinical development, which may cause actual performance and financial results in future periods to differ materially from any projections of future performance or results expressed or implied by such forward-looking statements. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. For additional disclosure regarding these and other risks faced by the Company, see the disclosure contained in the Company’s public filings with the US Securities and Exchange Commission (the “SEC”), including in the Company’s prospective filed with the SEC on September 21, 2020, as well as subsequent filings and reports filed with the SEC. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. Although we believe the expectations reflected in such forward-looking statements are reasonable, we can give no assurance that such expectations will prove to be correct. Accordingly, readers are cautioned not to place undue reliance on these forward-looking statements. No representations or warranties (expressed or implied) are made about the accuracy of any such forward-looking statements. 1

© COMPASS Pathways 2021 COMPASS’s leadership team George Goldsmith Chairman, CEO and Co-founder Ekaterina Malievskaia, MD Chief Innovation Officer, Co-founder Lars Wilde President, Chief Business Officer, Co-founder Piers Morgan Chief Financial Officer Nate Poulsen General Counsel and Head of Legal, IP, and Licensing Marco Mohwinckel Chief Commercial Officer Trevor Mill Chief Development Officer Tracy Cheung Chief Communications Officer 2 Sue Stansfield, PhD Senior Vice President, Clinical Operations Stephen Schultz Senior Vice President, Investor Relations Greg Ryslik, PhD Senior Vice President, Data Science, Machine Learning and Digital Health Research Gary Gilmour, DPhil Vice President, Preclinical Research Steven Levine, MD Vice President, Patient Access Sarah Bateup, Prof Doc Head of Therapy Research and Training Dr Stephen Wright Senior Scientific Advisor Emilio Arbe, MD Interim Clinical Sciences Director

© COMPASS Pathways 20213 3 Everyone has a S T O R Y

© COMPASS Pathways 2021CONFIDENTIAL | © COMPA S Pathways 20 We are a mental health care company 4 Source: 1. Depression and Other Common Mental Disorders: Global Health Estimates and Cleare, A. et al - 2015 -Evidence-based guidelines for treating depressive disorders with antidepressants: A revision of the 2008 British Association for Psychopharmacology guidelines. These sources state that 1/3 of those suffering with major depressive disorder (MDD) are estimated to be TRD. Therefore, we approximated 100 million from 320 million people with MDD 2. Halberstadt and Geyer – 2011 • Significant unmet need: 100m people1 with treatment-resistant depression (TRD) • Committed to transforming the patient experience Dedicated to accelerating patient access to evidence-based innovation in mental health care • COMP360 designated a FDA Breakthrough Therapy for TRD • Completed phase I healthy volunteers trial, largest psilocybin therapy clinical trial to date • Phase IIb ongoing, with 216 patients expected to have completed the trial by late 2021 • Planned expansion into additional indications Developing COMP360 psilocybin therapy for TRD • COMP360 differentiated mechanism of action, activating the 5HT2A receptor2 • Signals from academic studies have shown that psilocybin therapy can improve outcomes for patients • IP strategy combining patent protection with regulatory and market exclusivity Driven by science and rigour

© COMPASS Pathways 2021 Transforming the patient experience in mental health care Our vision A world of mental wellbeing Health systems and payer partnerships Value-based models, real-world evidence Innovative care delivery models Centres of Excellence, digital technologies FDA Breakthrough Therapy designation for COMP360 in TRD; New indications and compounds in development

© COMPASS Pathways 2021 6 TRD treatment pathway: significant unmet need for 100 million patients Treatment pathway stage New onset depression Major depressive disorder (MDD) Persistent depression Major depressive disorder (MDD) Treatment-resistant depression (TRD) Line of therapy Estimated no of patients (worldwide) 320 million 200 million 100 million (~33% of total) Available treatments • Antidepressants • Psychological interventions, eg CBT* • Antidepressants • Antidepressant combinations • Psychological interventions • Antidepressants • Augmentation therapy (antidepressants, mood stabilisers, anticonvulsants, atypical antipsychotics, esketamine) • Ketamine • Somatic therapy (rTMS*, tDCS*, ECT*, DBS*) • High-intensity psychological interventions % relapse 60-70% 50-75% 80-90% Note: *CBT = cognitive behavioural therapy; rTMS = repetitive transcranial magnetic stimulation; tDCS=transcranial direct current stimulation; ECT=electroconvulsive therapy; DBS=deep brain stimulation First line Second line Third line + Source: Hasler et al, 2004 - Acute psychological and physiological effects of psilocybin in healthy humans: a double-blind, placebo-controlled dose effect study

© COMPASS Pathways 2021 We need a new treatment model in TRD Source: WHO (2017); Depression Therapeutics by David Thomas and Chad Wessel, Bio Industry Analysis (2019); Johnston KM, Powell LC, Anderson IM, Szabo S, Cline S (2018). The burden of treatment- resistant depression: a systematic review of the economic and quality of life literature. Journal of Affective Disorders Note: TRD = treatment-resistant depression; MDD = major depressive disorder; 1. DALY = disability-adjusted life years; 2. Indirect costs are associated with the expenses incurred from the cessation or reduction of work productivity due to morbidity and mortality; 3. Accounting for comorbid physical and psychiatric conditions Depression: leading cause of disability worldwide Depression: burden on health systems2 TRD: increased economic and societal costs • MDD estimated to account for 7.5% of years of life lost due to disability globally, as defined by DALYs1 • Approx sevenfold increase in suicide rate for TRD patients compared with non-TRD MDD patients • US annual cost of depression: >$200 billion3 • A large proportion can be attributed to direct costs (eg outpatient and inpatient medical services and pharmaceutical services) • US medical costs for TRD patients are ~2-3x costs for non-TRD MDD patients • TRD patients have ~2x inpatient visits relative to non-TRD MDD patients • Average US annual healthcare cost between $17-25k per TRD patient per year Need for a new treatment paradigm New mechanisms of action Fewer side effects Rapid-acting and durable response 7
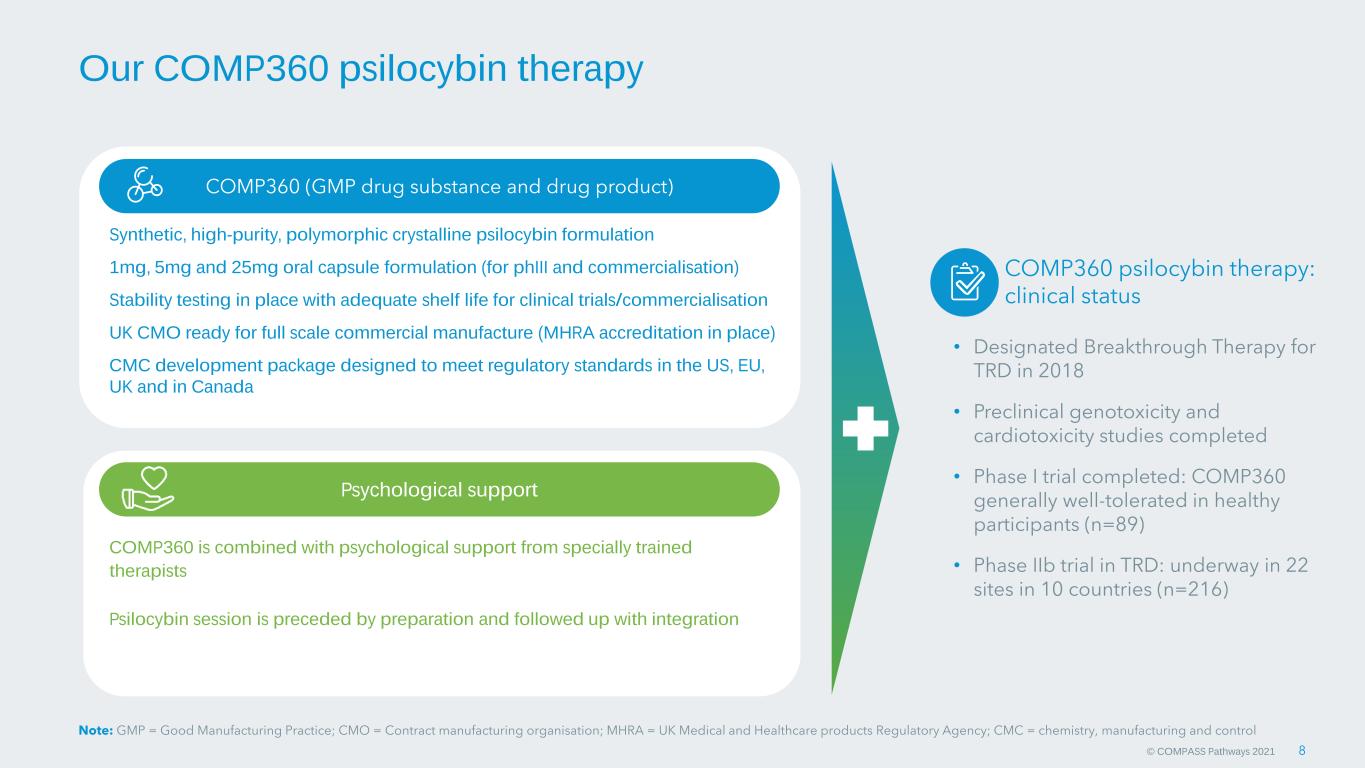
© COMPASS Pathways 2021 COMP360 psilocybin therapy: clinical status • Designated Breakthrough Therapy for TRD in 2018 • Preclinical genotoxicity and cardiotoxicity studies completed • Phase I trial completed: COMP360 generally well-tolerated in healthy participants (n=89) • Phase IIb trial in TRD: underway in 22 sites in 10 countries (n=216) Our COMP360 psilocybin therapy COMP360 (GMP drug substance and drug product) Psychological support COMP360 is combined with psychological support from specially trained therapists Psilocybin session is preceded by preparation and followed up with integration 8 Synthetic, high-purity, polymorphic crystalline psilocybin formulation 1mg, 5mg and 25mg oral capsule formulation (for phIII and commercialisation) Stability testing in place with adequate shelf life for clinical trials/commercialisation UK CMO ready for full scale commercial manufacture (MHRA accreditation in place) CMC development package designed to meet regulatory standards in the US, EU, UK and in Canada Note: GMP = Good Manufacturing Practice; CMO = Contract manufacturing organisation; MHRA = UK Medical and Healthcare products Regulatory Agency; CMC = chemistry, manufacturing and control

© COMPASS Pathways 2021 Clinical signals: early indicators from academic-sponsored trials show rapid reductions in symptoms in TRD and other mental health conditions 9 2011 2016 2016 2016, 2018 UCLA1 Grob et al n = 82 New York University Ross et al n = 232 Johns Hopkins Griffiths et al n = 462 Imperial College Carhart-Harris et al n = 193 2020 Johns Hopkins Griffiths et al n = 244 Significant reduction in BDI scores at six months, compared with baseline Effect size not reported p-value = 0.03 Significant reduction in HADS-D scores at 26 weeks post-dose 2, compared with baseline Effect size (Cohen’s d): niacin-first group = 0.66 psilocybin-first group =0.81 p-value < 0.05 Significant reduction in GRID-HAMD scores at six months across both groups, compared with baseline Effect size (Cohen’s d) = 2.98 p-value < 0.001 Significant reduction in GRID- HAMD scores in Immediate Treatment group at one and four weeks, compared with Delayed Treatment group Effect size (Cohen’s d): 1-wk = 2.2 4-wk = 2.6 p-value = <0.001 (1-wk and 4-wk) Significant reduction in QIDS score at six months, compared with baseline Effect size (Cohen’s d) = 1.6 p-value = 0.0035 Change in mean depression score from Baseline to 4-wk Existential distress Existential distress Existential distress Major depressive disorder Treatment-resistant depression Note: 1. UCLA = University of California, Los Angeles; 2. n denotes the number of patients who completed the relevant disclosed timepoint; 3. Denotes the number of patients for whom data is shown in the bar graph. A total of 19 patients completed six months follow-up; 4. Denotes the number of patients who completed both administration sessions and 1-wk and 4-wk post-session visits. All charts have been recreated from information provided in relevant papers. None of these studies used COMP360 0 5 10 15 20 25 Base- line 1 week 2 weeks 3 weeks 5 weeks 3 months 6 months QIDS (Depression)

© COMPASS Pathways 2021 Psilocybin therapy: potential benefits for patients, clinicians and payers Rapid and sustained relief A meaningful patient experience A sense of agency and empowerment Reduction in total cost of care Lower healthcare resource utilisation Increased productivity, reduced absenteeism Potential patient benefits Potential economic benefits 10

© COMPASS Pathways 2021 Psilocybin is a psychoactive substance 11 An active ingredient in some species of mushrooms Established knowledge of subjective effects A generally well-tolerated serotonergic psychedelic Psilocybin molecule

© COMPASS Pathways 2021 COMP360 mechanism of action 12 Source: 1. Halberstadt et al (2011); 2. Lopez-Gimenez et al (2018); 3. Vollenweider et al (1999); 4. Sakashita et al (2015); 5. Carhart-Harris et al (2012a); 6. Petri (2014); 7. Ly et al (2018) Note: *5-HT2A = 5-hydroxytyryptamine 2A; DMN = default mode network; mPFC = medial prefrontal cortex 2. Altered extracellular release of dopamine3,4 and leading to enhanced positive mood 3. Downregulation of the default mode network, or DMN*5, and de-synchronisation of cortical activity as well as the emergence of new patterns of functional connectivity across the brain6 4. Sustained cellular changes leading to neuroplasticity7 and “window of opportunity” for therapy 1. Stimulation of 5-HT2A receptors1 results in downstream cascades via G- protein signalling2 Modulation of cortical and limbic systems via 5-HT2A receptors Prefrontal cortex Posterior cingulate cortex Cortical system Amygdala Hippocampus Limbic system Pyramidal neuron Psilocin (metabolite of psilocybin) 5-HT2A receptor* NH OH N

© COMPASS Pathways 2021 Simplified visualisation of the acute changes in brain network connectivity 13 Placebo Psilocybin Brain network alterations may indicate the emergence of novel patterns of connectivity, following downregulation of the DMN Note: Figure adapted from Petri et al, 2014; study analysed fMRI (functional magnetic resonance imaging) data from healthy volunteers to compare resting-state functional brain connectivity after intravenous infusion of placebo and psilocybin Source: Petri, 2014 - Homological scaffolds of brain functional networks 13

© COMPASS Pathways 2021 Phase I safety and feasibility trial – data published in December 2019 Note: Study run in conjunction with Institute of Psychiatry, Psychology & Neuroscience, King’s College London Source: 'Eriksson - 2020 - Psilocybin therapy for treatment-resistant depression' and also 'COMPASS Pathways COMP 002 Safety Tables revised 2019-11-05' see table 14.3.1.2 • Largest randomised controlled study of psilocybin completed, in 89 healthy volunteers • COMP360 psilocybin was generally well-tolerated with no serious adverse events • No clinically-relevant negative effects on cognitive and emotional functioning • Feasibility of simultaneous administration to up to six people, with 1:1 support • Clinical training for phase IIb trial therapists 14

© COMPASS Pathways 2021 COMP360 induced psychedelic experiences that correlate with therapeutic effect 15 a. Ranked by incidence in the 25mg psilocybin group b. Includes auditory, gustatory, olfactory, tactile and visual hallucinations Most frequently reported AEs* (MedDRA Code) a in our phase I trial with healthy volunteers Mood altered AEs ranked by incidence in the 25mg psilocybin group Note: *AE = adverse event; MedDRA = Medical Dictionary for Regulatory Activities

© COMPASS Pathways 2021 Majority of adverse events resolved on day of administration, with a median duration of one day 16 Of all AEs, 68% reported as starting and resolving on the day of administration The median duration of AEs in all treatment arms across the 12-week trial was one day Most frequent AEs: onset and duration by treatment arm in our phase I trial

© COMPASS Pathways 2021 Integration 17 Psilocybin therapy: described by most patients in one study as being among the top five most meaningful experiences of their lives1 Source: 1.Griffiths et al (2016) Preparation Psilocybin session • Establish therapeutic alliance • Demonstrate and practise self- directed inquiry and experiential processing • Online preparation platform to remind patients what to expect and how to prepare • Supported by therapist and assisting therapist throughout 6-8 hour session • Room designed for non-clinical, calming atmosphere • Specially-designed music playlist, eyeshades to help focus internally • Patients often experience sense of connectedness, emotional breakthrough and acceptance • Therapists help patients process the emotional and physical experiences facilitated by psilocybin • Generate insights that can lead to cognitive and behavioural changes • Patients often experience a sense of agency and a separation from their symptoms, and report feeling empowered to make changes in their lives

© COMPASS Pathways 2021 COMPASS solutions in development Patient preparation platform Online therapist training and learning platform AI-assisted therapist feedback and monitoring Research Analyse digital biomarker data with the goal of predicting relapse and modelling disease course Develop technologies to augment or complement our therapies Developing and researching technology applications to improve the safety, efficacy and accessibility of our therapy 18 Note: AI = augmented intelligence Building a strong in-house team Greg Ryslik - SVP Data Science, Machine Learning and Digital Health Research • Former Chief Data Officer at Celsius Therapeutics; VP of Data Science at Mindstrong Health; Service Data Science Lead at Tesla Motors Bob Dougherty - VP, Digital Health Research • Former VP of Research at Mindstrong Health; Research Director at the Stanford Center for Neurobiological Imaging • Published 50+ peer-reviewed articles in psychology and neuroscience Sarah Bateup – Head of Therapy Research and Training • Former Chief Clinical Officer at Ieso Digital Health • Top 100 Business Leaders Award (2019); top 50 women in healthcare leadership (2018)

© COMPASS Pathways 2021 A rigorous therapist training programme 19 Note: TRD = treatment-resistant depression; 1. Development and Evaluation of a Therapist Training Program for Psilocybin Therapy for Treatment-Resistant Depression in Clinical Research, Sara J Tai, Elizabeth Nielson, Molly Lennard-Jones, Riikka Ajantaival, Rachel Winzer, William A Richards, Frederick Reinholdt, Brian D Richards, Peter Gasser, Ekaterina Malievskaia, Frontiers in Psychiatry, February 2021 Manualised methodology drawing from evidence-based psychotherapeutic approaches Ongoing clinical supervision and professional development In-person training Applied clinical training Online learning platform Developed with leading experts in mental health and psychedelic research Used to train therapists in our phase IIb trial of COMP360 psilocybin therapy for TRD Formal and scalable methodology for psychological support in psilocybin therapy Training programme will continue to evolve Programme details shared in a paper written jointly with academic researchers and published in peer-reviewed Frontiers in Psychiatry1 4 components

© COMPASS Pathways 2021 Phase IIb clinical trial: COMP360 psilocybin therapy for TRD Target enrolment of 216 patients; data expected late 2021 20 Primary endpoint ✧ Reduction of symptoms of depression as measured by MADRS* from Baseline to 3 weeks Secondary endpoint ✧ Proportion of responders who maintained ≥ 50% improvement in MADRS up to week 12 Weekly visits (V1a, V1b, etc) Screening (V1) D-1: Baseline (V2) Day 1 (V4) Week 3 (V7) Week 12 (EOS*, V10) D0: Psilocybin Session (V3) RANDOMISATION 1:1:1 3-6 weeks Day 0 Day 1 Week 3 Week 6 (V8) Remote visit Week 9 (V9) Remote visit Week 12 Week 1 (V5) Week 2 (V6) Week 1 Week 2 72 subjects 72 subjects 72 subjects 25mg psilocybin10mg psilocybin1mg psilocybin Note: *MADRS = Montgomery-Åsberg depression rating scale; EOS = end of study; TRD = treatment-resistant depression To date, two patients have experienced suspected, unexpected serious adverse reactions (SUSARs) in our phase IIb trial for TRD patients

© COMPASS Pathways 2021 Sites engaged for phase IIb TRD study have the reputation and capability to recruit patients 21 Site Country with engaged site Groningen London (2) Newcastle Barcelona (2) Aalborg DublinToronto New York City Baltimore Atlanta Houston San Diego (2) Stanford Bristol Utrecht Leiden Lisbon Czech Republic Berlin Psykiatrien i Aalborg Mølleparkvej University Medical Centre Groningen University Medical Centre Utrecht Charité - Universitätsmedizin Berlin National Institute of Mental Health Czech Republic Leiden University Medical Center Kings College London Institute of Psychiatry, Psychology & Neuroscience St Pancras Clinical Research Sant Joan De Deu Serveis De Salut Mental Institute Hospital del Mar of Medical Research (IMIM) Centro Clinico Champalimaud Unidade de Neuropsiquiatria University of California, San Diego Kadima Neuropsychiatry Institute Stanford University University of Texas Health Science Center Emory University School of Medicine Wolfson Research Centre, Northumberland, Tyne and Wear NHS Foundation Trust Avon and Wiltshire Mental Health Partnership NHS Trust Tallaght University Hospital University of Toronto Centre for Addiction and Mental Health New York State Psychiatric Institute Sheppard Pratt Health System Note: TRD = treatment-resistant depression

© COMPASS Pathways 2021 Our clinical development programme for COMP360 psilocybin therapy in TRD Getting ready for phase III 22 Safety and feasibility 89 healthy volunteers Phase IIIPhase IIPhase I Status Safety and efficacy Primary endpoint: change in MADRS 216 patients, 22 sites in EU, US, Canada Long-term follow-up study of participants from phase IIb trial Psilocybin as adjunct to SSRI aims to confirm that COMP360 is best administered as monotherapy Completed Dec 2019 Recruiting; results expected end 2021 Recruiting; results expected end 2021 Recruiting; results expected end 2022 Note: SSRI = Selective serotonin reuptake inhibitors; TRD = treatment-resistant depression

© COMPASS Pathways 2021 COMPASS Pathways: leader in psilocybin therapy research 23 IISs using COMP360: signal-generating, exploratory studies looking at indications in areas of unmet need Note: IIS = investigator-initiated studies, MDD = major depressive disorder, TRD = treatment-resistant depression We do not sponsor investigator-initiated studies, some of which use their own protocols and study design. We encourage the open publication of all associated findings from any study or trial using COMP360 • COMPASS has pending patent applications that include the indications listed • COMPASS has the right to exclusively license new IP generated through these studies • Studies may provide signals that we can explore further and expand in a portfolio approach to different indications MDD Anorexia University of Zurich Severe TRD Sheppard Pratt Body dysmorphic disorder Columbia University MDD comparative mechanism of action Imperial College London Aquilino Cancer Center Chronic cluster headache University of Copenhagen UC San Diego Bipolar disorder II Sheppard Pratt TRD King’s College London Suicidal ideation Sheppard Pratt Autism King’s College London

© COMPASS Pathways 2021 Our first Centre of Excellence, in collaboration with Sheppard Pratt, to accelerate research in a range of mental health illnesses One of the world’s leading research institutions in mental health and one of the top psychiatric hospitals in the US A leader in clinical service delivery, supporting 70,000+ patients in 42 states and 19 countries Part of the COMPASS phase IIb TRD clinical trial, and long-term follow-up study Also using COMP360 psilocybin in two IISs in psilocybin therapy for severe TRD and for bipolar type II depression The CoE will be used to: • Conduct clinical trials in psilocybin therapy for a range of mental health illnesses • Train and certify therapists • Prototype digital solutions to improve patient experience Centre of Excellence (CoE): a research facility and innovation lab to model the clinic of the future A world-class institution Note: TRD = treatment-resistant depression; IIS = investigator-initiated study A recognised team led by Scott Aaronson MD, Director of Clinical Research at Sheppard Pratt, distinguished fellow of the American Psychiatric Association and fellow of the American College of Psychiatrists The Sheppard Pratt campus 24

© COMPASS Pathways 2021 Exploring the potential of COMP360 therapy for major depressive disorder in cancer with Maryland Oncology Hematology at the Aquilino Cancer Center Signal-generating study (IIS) Major depressive disorder in cancer - an unmet medical need Purpose-built facility A 30-patient, open-label study to test the safety and feasibility of psilocybin therapy to treat depression in cancer patients FDA-approved protocol includes key features of a scalable delivery model, with simultaneous administration and 1:1 patient support under lead therapist supervision Study began in Q4 2020 with data expected in Q4 2021 New treatment space at the Aquilino Cancer Center (near Washington DC) co-designed with COMPASS for simultaneous delivery of psilocybin therapy Recognised unmet need and patient population Signal-generating study will inform later stage development path Development path to be designed in collaboration with regulators and stakeholders Note: IIS = investigator-initiated study 25

© COMPASS Pathways 2021 COMPASS Pathways Discovery Center A collaboration among world-leading scientists and institutions 26 John D McCorvy, leading 5-HT receptor pharmacologist and expert on GPCR signalling • Assistant Professor, Department of Cell Biology, Neurobiology and Anatomy, Medical College of Wisconsin Development of optimised novel psychedelic compounds targeting the 5-HT2A receptor COMPASS is a joint owner and exclusive licensee for all new compounds generated University of California San Diego (San Diego, California) University of the Sciences (Philadelphia, Pennsylvania) Medical College of Wisconsin (Milwaukee, Wisconsin) Adam Halberstadt, expert in behavioural psychopharmacology • Associate Professor, Department of Psychiatry, University of California San Diego Jason Wallach, leading chemist and pharmacologist in psychedelic and dissociative drugs • Assistant Professor of Pharmaceutical Sciences, University of the Sciences Note: GPCR = G protein-coupled receptor

© COMPASS Pathways 2021 Comprehensive preclinical program - COMP360 psilocybin in a broad range of indications with pending patent applications 27 Preclinical research has been conducted in the following indication areas: Ongoing confidence in these biological substrates builds preclinical extrapolations to the following indications Inflammatory bowel disease Post-traumatic stress disorder Traumatic brain injury Inflammation Chronic pain Binge eating disorder Post-partum depression Fibromyalgia Parkinson’s diseaseAlzheimer’s disease Cluster headache Anorexia nervosa Body dysmorphic disorder Social anxiety disorderMigraine Attention deficit hyperactivity disorder Sleep wake disorders Epilepsy Autistic spectrum disorder Generalised anxiety disorder Obsessive-compulsive disorder Panic disorderBulimia nervosa Stroke

© COMPASS Pathways 2021 COMP360 commercial exclusivity strategy First US patent granted in December 2019 • Claims directed to methods of treating drug-resistant depression with high-purity polymorphic crystalline psilocybin • Petition for Post Grant Review was dismissed on merits in August 2020 28 European patents granted/registered • German utility model (March 2020): covering forms of crystalline psilocybin, its use in medicine and methods of synthesis • First UK patent (May 2020): includes two independent method of manufacture claims, and product-by-process and formulation claims • Second UK patent (July 2020): includes claims covering crystalline psilocybin, pharmaceutical formulations, medical uses, and a method of manufacturing Multiple related applications pending • To expand claim scope • To extend coverage in over 20 additional countries/regions Three PCT applications and Taiwanese application pending • Additional formulations, administration, therapeutic and digital supports, combination treatments, methods of treating variety additional indications • Additional indications include: anxiety disorders, headache disorders, eating disorders, neurocognitive disorders, autism, epilepsy, inflammation, ADHD*, substance use disorders, inflammatory bowel disease, stroke, ALS*, multiple sclerosis, anti-social personality disorder, pain, sleep-wake disorders, and bipolar type II depression COMP360 can be registered as NCE*/NAS* • Possibility of full patent and regulatory exclusivity • Data protection, up to • 8-11 years (EU) • 5-7.5 years (US) Reschedule COMP360 psilocybin • Upon approval by FDA, COMP360 psilocybin could be rescheduled by DEA Patent strategy Note: *NCE = new chemical entity; NAS = new active substance; ADHD = attention deficit hyperactivity disorder; ALS = amyotrophic lateral sclerosis Regulatory strategy

© COMPASS Pathways 2021 Achieving broad patient access • Early scientific advice with key payer-experts and HTAb* • US reimbursement and coding strategy • Real-world evidence - data access agreements • Centres of Excellence • Prospective payer-focused trials • Potential franchise model Comprehensive and payer-relevant evidence generation plan Strategic partnerships with payers, health systems and clinic networks • Therapist training services and partnerships • Treatment centre activation services • Digital solutions – companion apps for prediction and prevention Differentiated and modular commercial offering 29Note: *HTAb = Health Technology Assessment bodies; Centre of Excellence prototype designs by Gensler (architecture, design, planning and consulting firm) Prototype design Centre of Excellence treatment room Prototype design Centre of Excellence post-treatment space

© COMPASS Pathways 2021 Financial overview Cash and cash equivalents at 30 September 2020 • $196.5 million IPO raise • $146.6 million1 Issued shares • 35,930,331 Covering analysts • Ritu Baral, Cowen • Josh Schimmer, Evercore • Esther Hong, Berenberg • Sumant Kulkarni, Canaccord Genuity • Patrick Trucchio, HC Wainwright & Co 30 Note: 1. IPO effective 18 September 2020; Greenshoe fully exercised

© COMPASS Pathways 2021 31 A strong and growing team

© COMPASS Pathways 2021 Backed by high calibre boards Board of directors George Goldsmith Ekaterina Malievskaia, MD Florian Brand Jason Camm Annalisa Jenkins, MBBS Thomas Lönngren Linda McGoldrick Robert McQuade, PhD David Norton Scientific advisory board Prof David Nutt, MD, PhD Gül Dölen, MD, PhD Thomas Insel, MD Prof Diego Pizzagalli, PhD Prof Augustus John Rush, MD Prof Alan Schatzberg, MD Paul Summergrad, MD Kirk Rutter (Patient Advisor) 32Note: Companies listed reflect prior and present relevant experience

© COMPASS Pathways 2021 Pioneering the development of a new model of psilocybin therapy 2021 Anticipated milestones Establish first Centre of Excellence Phase IIb trial: data expected late 2021 Further senior appointments Further partnerships and collaborations Data published from IISs using COMP360 Expand current IP portfolio with additional patent grants Evolve data and technology strategy 33 Key achievements $146.6m raised in September IPO; $80m raised in Series B Breakthrough Therapy designation for COMP360 in TRD Phase I healthy volunteers trial completed Phase IIb clinical trial making steady progress Patent awards in US, UK, Germany Experienced leadership team, board of directors, scientific advisory board; leadership team and board strengthened with recent hires Preclinical studies in new indications; Discovery Center launched Additional trials underway in TRD programme COMP360 used in multiple IISs exploring range of indications FDA approved request for 1:1 therapist patient ratio and online therapist training Aquilino Cancer Center launches psilocybin therapy study with simultaneous administration and 1:1 therapist support Note: TRD = treatment-resistant depression

© COMPASS Pathways 2021 I had such instant relief I could make up my mind about things … it lifted the fog of depression. The way I felt after, I have not felt with any medicine or therapy … I forgot what depression was. Quote from participant in Imperial College London psilocybin therapy study conducted by Carhart-Harris et al, 2016; image is representative and not of a patient

© COMPASS Pathways 2021 Stephen Schultz SVP, Investor Relations stephen.schultz@compasspathways.com +1 401-290-7324
